Standard entropy
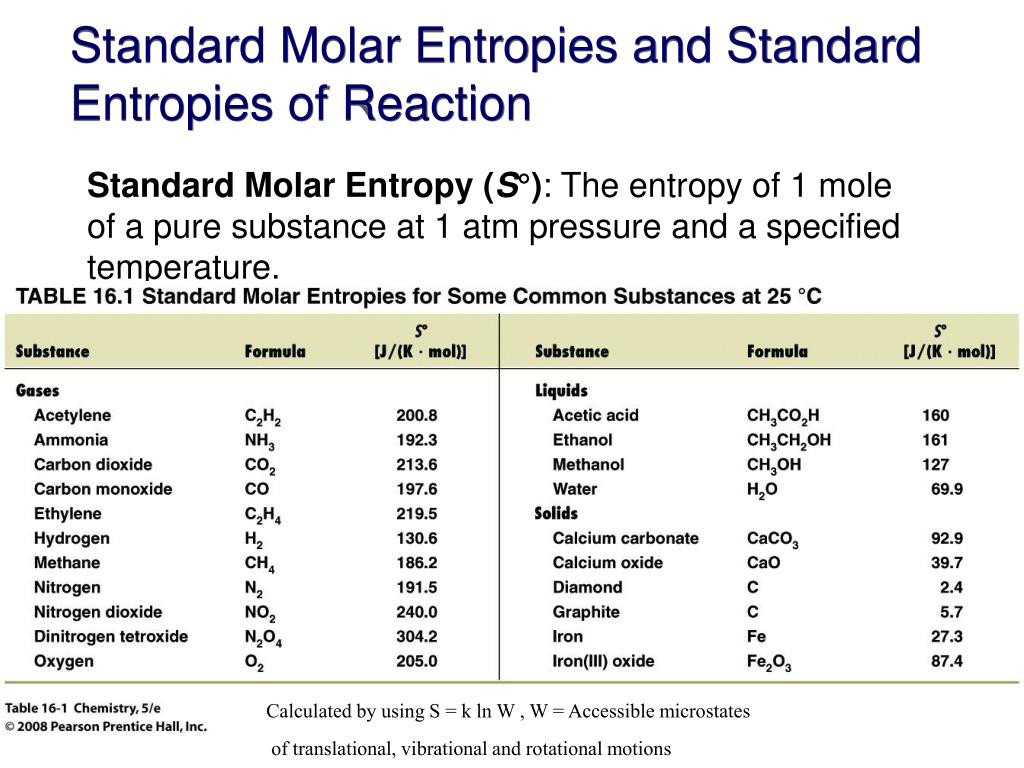
Second, this paper, despite its financial focus, presents potential reasons for preferring using entropy to standard deviation. induction agents and volatile anaesthetics like propofol and sevoflurane and overall was found comparable to the current clinical gold standard bispectral index (BIS). First, this discussion is useful to see why your statements 'higher the standard deviation, lesser the entropy' and 'lesser the SD, more the entropy' original style preserved are incorrect. Generally, entropy has been studied for quantification of anaesthetic drug effect for various GABA-ergic i.v. By calculating two distinct values for the EEG dominated part of the spectrum (state entropy, SE) and the total spectrum (response entropy, RE), the M-Entropy module claims to provide useful information regarding the cortical state of the patient as well as an indirect measure of adequacy of analgesia. In chemistry, standard molar entropy is defined as the entropy of 1 mole, or gram molecule, of matter at a standard atmospheric pressure of 14.7 lbs/inch 2 (101.3 kPa) and a given temperature. Various entropy algorithms have been used in clinical studies, but until now a commercially available monitor exists only for spectral entropy. The definition of standard entropy has slightly different meanings depending on the field of science to which it is being applied. Our procedures are simple, robust, and reliable and can be used by specialists and nonspecialists alike.The concept of entropy, originally derived from thermodynamics, has been successfully applied to EEG analysis. A pure element in its standard state has a standard enthalpy of formation of zero. Coupled with our published procedures, which relate volume to other thermodynamic properties via lattice energy, the correlation reported here complements our development of a predictive approach to thermodynamics and ultimately permits the estimation of Gibbs energy data. The standard enthalpy of formation of a compound is the enthalpy change that occurs when 1 mole of the compound is formed from its constituent elements in their standard states. The regression lines pass close to the origin, with the following formulas: For inorganic ionic salts, S degrees 298 /J K(-)(1) mol(-)(1) = 1360 (V(m)()/nm(3) formula unit(-)(1)) + 15 or = 2.258 + 15. The approach can also be extended to estimate entropies for hypothesized materials. The standard entropy of the H+(aq) ion is defined to be 0. V(m)() can be obtained from a number of possible sources, or alternatively density, rho, may be used as the source of data. Standand Enthalpies of Formation & Standard Entropies of Common Compounds. q o (ion) have been found to correlate linearly with the standard ionic entropies of hydration for the 38 ions investigated. It is shown here that formula unit volume, V(m)(), can be employed for the general estimation of standard entropy, S degrees 298 values for inorganic materials of varying stoichiometry (including minerals), through a simple linear correlation between entropy and molar volume.
STANDARD ENTROPY FULL
Standard absolute entropies of many inorganic materials are unknown this precludes a full understanding of their thermodynamic stabilities.
